Shane Eckenrode
Software Developer & Engineer
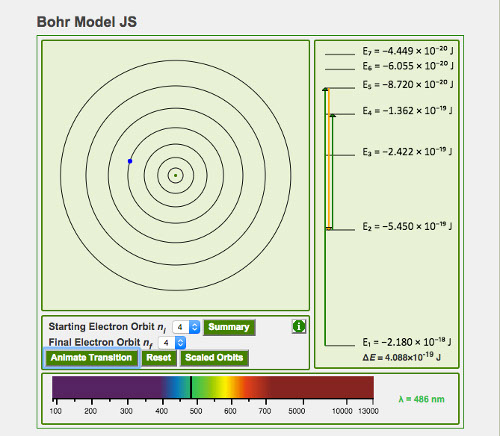
The Bohr Model project is a JavaScript application developed as a more mobile-friendly solution from it's original design. The original applet was created using the Java language that required a plugin for use on the chemistry eBook website.
The model consists of 4 different drawing canvases with the main canvas showing the Hydrogen atom with 7 possible orbitals for the electron to reside on. The electron is constantly animated around the orbitals.
There is a linear display which is used to draw linear arrows when the electron transitions to a new orbital. A secondary linear display can be displayed below the application which will show the orbits to scale and also draws linear arrows when a transition has occurred.
The last canvas shows the spectrum in relation to the energy which is either absorbed or emitted when the electron transitions to a new orbital. There is also a summary table pop-up window to store all of the calculated data when the electron is animated.
A user can animate the electron by selecting a starting and final orbital. When the animate button is clicked, a photon will react to the electron accordingly and move the electron to it’s selected final destination. The other canvases will also animate to depict the changes resulting from the transition.
This model provides students with the opportunity to experiment with the Bohr Model concepts and to gain a better understanding of the inner workings of the different electron transitions.